Project 1
Swathi Kiran, Ph.D., CCC-SLP and David Caplan, M.D., Ph.D. study recovery of naming ability. Treatment focuses on naming objects or things within semantic categories based on their meaning and the features that connect them. Training atypical items within categories (e.g., ostrich in the category of birds) improves these words as well as more typical items within category (e.g., robin). This project examines recovery of naming and other language abilities as well as the effects of treatment on brain processing.
Dr. Kiran is an Associate Professor in the Department of Speech, Language, and Hearing Sciences at Boston University. She is an expert and leading authority on naming deficits in aphasia.
Dr. Caplan is Professor of Neurology at Harvard University and Director of Cognitive-Behavioral Neurology at the Massachusetts General Hospital. He is an expert and leading authority on the brain and language.
Recent Projects
Treatment, generalization and selectivity. During this experiment, 27 people with aphasia (PWA) (17 male; age range=42-80 years) received a typicality-based Semantic Feature Analysis (SFA) treatment and generalization was examined across three levels: untrained related items, semantic/phonological processing tasks, and measures of global language function. Additionally, 10 PWA (10 male; age range=39-79 years) served as natural history controls. Our results showed that the likelihood of naming trained items was significantly higher than for monitored items for the treated PWA group. The treated PWA group also showed significant post-treatment gains on semantic/phonological processing tasks and standardized assessments, therefore confirming the effectiveness of the treatment.
Cognitive factors predicting recovery. Here, we have examined the influence of baseline cognitive abilities on recovery of anomia after treatment. Controlling for pre-treatment apraxia of speech and auditory comprehension impairment, regression models, explaining 57% and 62% of the variance, respectively, revealed executive functioning (EF) and visual short-term memory (ViSTM) to predict naming improvement and maintenance of gains. These findings indicate that strong pre-treatment executive function and visual short-term memory abilities result in greater improvements and maintenance of those improvements for individuals with chronic aphasia undergoing semantic feature-based naming therapy.
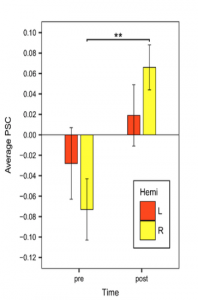
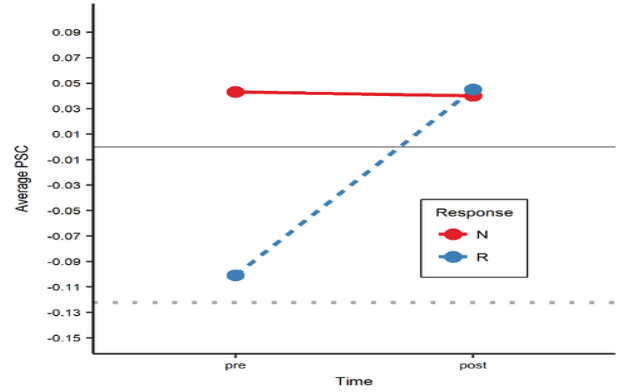
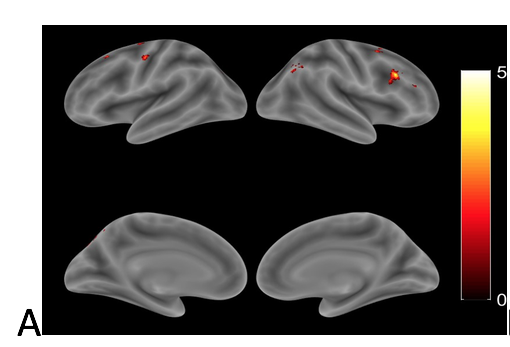
Neurovascular changes after treatment. In order to examine the neurovascular and cognitive changes associated with anomia treatment, an fMRI picture-naming task was used to examine 16 regions of interest in 26 patients with chronic aphasia, before and after 12 weeks of semantic naming treatment. Ten control patients, who did not receive treatment, and 17 healthy controls were also scanned. We found that, naming therapy resulted in a significant increase in cortical activation (see Figure 1), an effect that was largely driven by patients who responded most favorably to treatment (see Figure 2), as patients who responded less favorably (as well as those who did not receive treatment) had little change in activation over time. Relative to healthy controls, patients had higher pre-treatment activation in the bilateral inferior frontal gyri (IFG) and lower activation in the bilateral angular gyri; after treatment, they had higher activation in bilateral IFG, as well as in the right middle frontal gyrus. These results suggest that the predominant effect of beneficial naming treatment was an upregulation of traditional language areas and their right hemisphere homologues and, in particular, regions associated with phonological and semantic/executive semantic processing, as well as broader domain general functions.
In another analysis, we examined changes in activation during a semantic feature verification task as a function of the semantic based treatment in 19 healthy controls (mean age= 59.2 years) and 20 PWA. Figure 3a shows group changes in activation (post-treatment relative to pre-treatment) in left superior frontal gyrus (LSFG), left middle frontal gyrus (LMFG), right superior frontal gyrus (RSFG), right middle frontal gyrus (RMFG), right inferior frontal gyrus opercularis (RIFGop)¸ L Postcentral Gyrus, left superior occipital gyrus (LSOG), L Precuneus, R Angular Gyrus (RAG), right superior occipital gyrus (RSOG), after accounting for lesion volume.
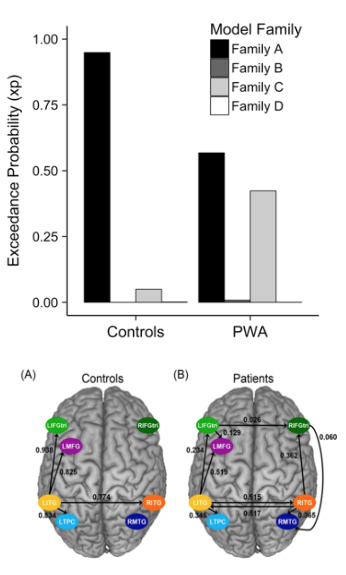
Effective connectivity after stroke. First, we investigated connectivity of the naming network using dynamic causal modeling (DCM) and found that PWA tended to rely on more domain-general (i.e., LMFG) rather than “classic” language regions (e.g., left inferior frontal gyrus (LIFG) and left middle temporal gyrus (LMTG)). To further understand to what extent damage to left hemisphere regions influenced effective connectivity patterns in both hemispheres, we examined PWA and healthy control performance on a semantic judgment task. Using fMRI and DCM, biologically-plausible models within four model families were created to correspond to potential neural recovery patterns, including Family A: Left-lateralized connectivity (i.e., no/minimal damage), Family B: Bilateral anterior-weighted connectivity (i.e., posterior damage), Family C: Bilateral posterior-weighted connectivity (i.e., anterior damage) and Family D: Right-lateralized connectivity (i.e., extensive damage) (Figure 4). As predicted, controls exhibited a strong preference for left-lateralized network models (Family A) whereas patients demonstrated a split preference between Families A and C. At the level of connections, controls exhibited stronger left intra-hemispheric task-modulated connections than did patients. These results suggest that lesion site partially (but not fully) predicted network connectivity patterns and language abilities.
Neural differences between responders and nonresponders. In an additional study, we examined if functional connectivity changes in PWA (N= 26) emerged as a function of treatment and if these changes were different from connectivity patterns in untreated patients (N=10) and healthy controls (N =17). Patients with aphasia had significantly lower connectivity than healthy controls at the start of the study (see Figure 5, row 1). After treatment, the number of abnormal connections decreased (column 1), suggesting a degree of normalization of connectivity in treated patients but not in untreated patients (column 2). Also, interregional connectivity normalized in patients whose naming accuracy improved the most after treatment (column 3), but remained abnormally low in patients who responded less favorably (column 4) or who did not receive treatment at all (column 2). From this we found that, responders’ pre-treatment network properties were more control-like than those of nonresponders and normalized after treatment. On the other hand, nonresponders’ showed normalization of measures of local processing but continued to have lower network strength and global efficiency than healthy controls after treatment. Therefore, patients who had the best behavioral response to treatment had more typical network properties from the outset and a greater capacity for normalization of interregional and global network connectivity than those who had poorer treatment outcomes.